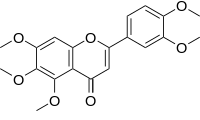
Sinensetin
 | |
 | |
| Names | |
|---|---|
| IUPAC name 3′,4′,5,6,7-Pentamethoxyflavone | |
| Systematic IUPAC name 2-(3,4-Dimethoxyphenyl)-5,6,7-trimethoxy-4H-1-benzopyran-4-one | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChEMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.230.396 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C20H20O7 |
| Molar mass | 372.36 g/mol |
| Hazards | |
| GHS labelling:[1] | |
Pictograms |  |
| Danger | |
Hazard statements | H301 |
Precautionary statements | P264, P270, P301+P310, P321, P330, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Sinensetin is a methylated flavone. It can be found in Orthosiphon stamineus[2] and in orange oil.[3]
References
- ^ GHS: Pubchem
- ^ Akowuah, G. A.; Ismail, Z.; Norhayati, I.; Sadikun, A. (2005). "The effects of different extraction solvents of varying polarities on polyphenols of Orthosiphon stamineus and evaluation of the free radical-scavenging activity". Food Chemistry. 93 (2): 311–317. doi:10.1016/j.foodchem.2004.09.028.
- ^ Steinke, Katrin; Jose, Elena; Sicker, Dieter; Siehl, Hans-Ullrich; Zeller, Klaus-Peter; Berger, Stefan (2013). "Sinensetin". Chemie in unserer Zeit (in German). 47 (3): 158–163. doi:10.1002/ciuz.201300627.
- v
- t
- e
Flavones and their conjugates
| Monohydroxyflavone | |
|---|---|
| Dihydroxyflavones | |
| Trihydroxyflavones | |
| Tetrahydroxyflavones | |
| Pentahydroxyflavones |
|
| O-methylated flavones |
|
| of apigenin | |
|---|---|
| of baicalein | |
| of hypolaetin |
|
| of luteolin |
- Giraldiin A and B
- Nepitrin
- Oroxindin
- Scutellarin
Theograndin I and II
 | This article about an aromatic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e












